BiP (Binding Protein)
BiP (Binding Protein)는 ER lumen의 major chaperone protein으로, hsp70 family에 속한다. Protein folding in ER에서 핵심적인 역할을 하며, misfolded protein을 인식하고 aggregation을 방지한다.
구조와 특성
Family
- Hsp70 family member
- 다른 이름: GRP78 (Glucose-Regulated Protein 78)
- Highly conserved across eukaryotes
Localization
- ER resident protein
- ER retention signal: KDEL sequence (C-terminus)
- Predominantly in ER lumen
- Stress 시 세포 표면으로도 이동 가능
Structure
- N-terminal domain: ATPase domain
- C-terminal domain: Substrate-binding domain
- Linker region: Domain communication
기능적 역할
1. Post-Translational Translocation
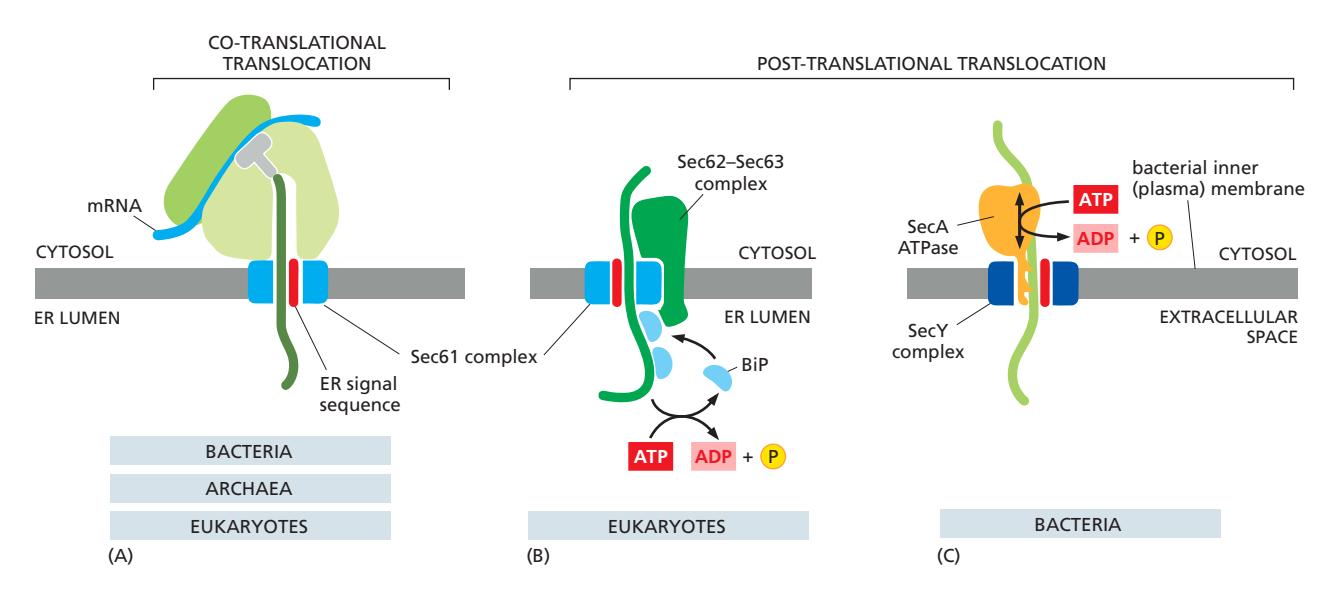
Pulling motor:
- TIM23 complex와 결합
- Matrix space에서 작용
- Emerging polypeptide에 결합
- ATP-dependent pulling force
Mechanism:
- Polypeptide가 TIM23을 통해 나타남
- BiP가 즉시 결합 (high affinity)
- ATP hydrolysis → Conformational change
- Pulling force 발생
- BiP release, substrate rebinding
- Cycle 반복
2. Co-Translational Assistance
- Nascent chain과의 상호작용
- Cotranslational folding 보조
- Premature folding 방지
3. Protein Folding Chaperone
Recognition:
- Exposed hydrophobic sequences
- Unfolded or partially folded protein
- Incorrectly assembled oligomeric complex
Prevention of aggregation:
- Hydrophobic region masking
- Protein-protein interaction 차단
- Solubility 유지
4. ER Retention
Incomplete assembly:
- Unassembled subunit 인식
- ER에 retention
- 완전한 assembly까지 유지
- Golgi로의 이동 방지
5. Quality Control Sensor
UPR regulation:
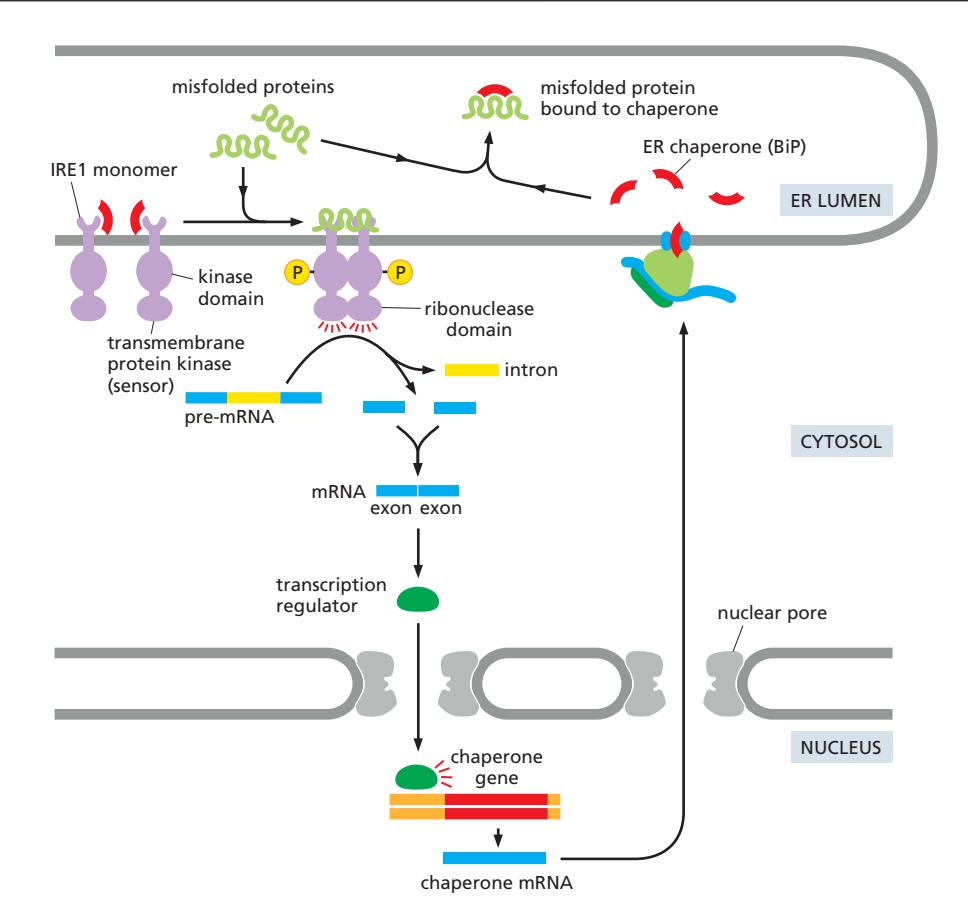
- IRE1, PERK, ATF6에 결합
- Normal condition: UPR sensor 억제
- Stress condition: Misfolded protein과 결합하기 위해 dissociate
- UPR sensor 활성화
ATP-Dependent Cycle
Mechanism
-
ATP-bound state:
- Low affinity for substrate
- Open conformation
- Rapid substrate exchange
-
ATP hydrolysis:
- Conformational change
- High affinity for substrate
- Tight binding
-
ADP-bound state:
- Substrate firmly held
- Folding attempt opportunity
-
Nucleotide exchange:
- ADP → ATP
- Return to low affinity
- Substrate release
Co-chaperones
Nucleotide exchange factors (NEFs):
- GRP170 (mammalian)
- Sil1 (yeast)
- ADP → ATP exchange 촉진
- BiP cycle 가속
J-domain proteins:
- ERdj family
- BiP의 ATPase activity 자극
- Substrate specificity 제공
- BiP를 특정 substrate로 recruit
다중 역할의 통합
Versatile Chaperone
- Translocation: Pulling motor
- Folding: Aggregation prevention
- Quality control: Misfolded protein detection
- Stress sensing: UPR regulation
Context-Dependent Function
- Location에 따라 다른 역할
- Partner protein에 따라 기능 변화
- Cellular state에 반응
Regulation
Expression
Constitutive:
- Basal level 항상 유지
- Essential function
Inducible:
- ER stress로 upregulation
- UPR에 의한 transcription 증가
- Glucose deprivation에 반응
Activity
Post-translational modification:
- Phosphorylation
- ADP-ribosylation
- Activity modulation
Co-chaperone:
- J-domain protein recruitment
- NEF availability
- Substrate specificity
BiP와 다른 Chaperone의 협력
PDI
- Disulfide bond formation
- BiP와 sequential action
- Coordinated folding
Calreticulin
- Glycoprotein-specific
- Parallel pathway
- Complementary recognition
ERp57
- Associated with calnexin/calreticulin
- Disulfide bond 관련
- Integrated quality control
질병과의 연관
Genetic Diseases
Marinesco-Sjögren syndrome:
- BiP co-chaperone (SIL1) mutation
- Cerebellar ataxia
- Cataracts
- Myopathy
Cancer
Tumor survival:
- BiP overexpression
- ER stress tolerance
- Chemotherapy resistance
- Therapeutic target
Neurodegenerative Diseases
Alzheimer’s, Parkinson’s:
- BiP dysregulation
- Protein aggregation
- ER stress
- Cell death
Experimental Tools
BiP as Marker
- ER stress indicator
- UPR activation marker
- Cell stress reporter
Functional Studies
- BiP depletion
- Dominant-negative mutant
- ATPase-deficient mutant
- Substrate-binding mutant
치료적 접근
Target for Therapy
Cancer:
- BiP inhibitor development
- ER stress sensitization
- Combination therapy
Protein Folding Diseases:
- BiP overexpression
- Chemical chaperone
- ER stress reduction